iPLEDGE Compliance Checklist
Why the Strict Controls Exist
To understand why the FDA requires such a rigid system, you have to look at the teratogenic effects of the drug. When a person is pregnant and takes isotretinoin, the results are often devastating. We aren't talking about minor complications; we're talking about abnormally shaped skulls, severe brain damage that affects both movement and cognition, and heart defects. Other common issues include cleft palates and the absence of ear canals. Because these risks are so severe, the program operates on a zero-tolerance policy: no female patient starts therapy if pregnant, and no patient becomes pregnant during treatment. There are no extensions, no exceptions, and no "grace periods" for these safety rules.Who Needs to Do What?
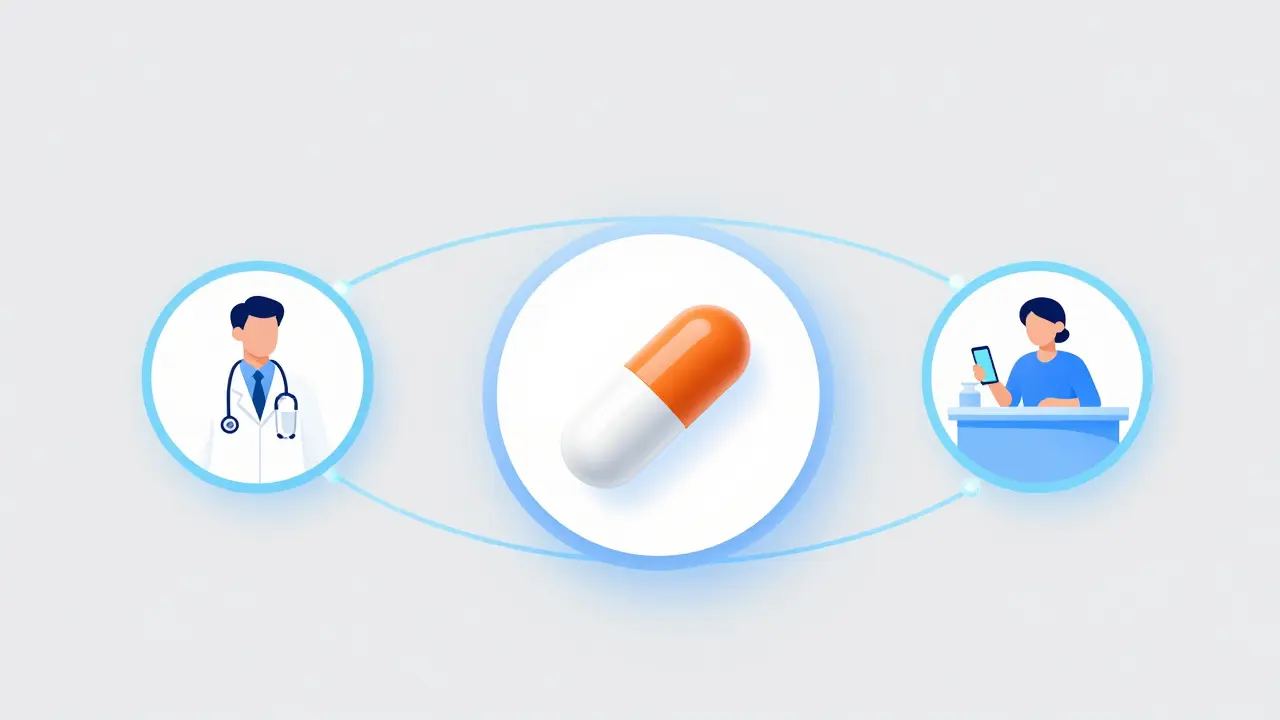
Getting started with Isotretinoin (often known by brands like Claravis, Amnesteem, or Zenatane) requires a coordinated effort. You can't just walk into a pharmacy with a script; everyone involved must be registered in the system.- The Prescriber: Your dermatologist must be registered and have completed specific FDA-mandated training. They are responsible for enrolling you in the system and verifying your monthly tests.
- The Pharmacy: Not every pharmacy handles these drugs. Your pharmacist must have an active iPLEDGE account to dispense the medication.
- The Patient: You must complete online education modules-which usually take about 30 minutes-and sign electronic agreements acknowledging the risks before you can be enrolled.
Breaking Down the Monthly Requirements
Your experience with the program depends heavily on whether you are categorized as "capable of pregnancy" or "not capable of pregnancy." The requirements for those capable of pregnancy are significantly more intensive.| Requirement | Patients Capable of Pregnancy | Patients Not Capable of Pregnancy |
|---|---|---|
| Initial Testing | Two negative pregnancy tests (second one within 1-3 days of start) | None required |
| Monthly Testing | One negative pregnancy test every month | None required |
| Contraception | Two forms of contraception used simultaneously | N/A |
| Counseling | Monthly electronic acknowledgment of risks | Enrollment-only acknowledgment |
| Verification | Strict monthly verification by prescriber | General compliance with guidelines |

The 2023 Updates: A Breath of Fresh Air
For years, patients and doctors complained that the program was "excruciatingly difficult." One of the biggest pain points was the 19-day "lockout" period-if you didn't pick up your meds within a narrow window, you could be locked out of the system for nearly three weeks. Thankfully, the FDA rolled out major changes on November 30, 2023, to make the process more human. First, the lockout period is gone. You no longer have to worry about a rigid 7-day window causing a massive gap in your treatment. Second, the program now allows for home pregnancy testing. In the past, you had to visit a lab or clinic every single month. Now, as long as your doctor has a verification protocol to prevent cheating, you can test at home and report it. This has been a game-changer for people living in rural areas who used to spend hours driving to a clinic just for a five-minute test. Additionally, the requirements for patients not capable of pregnancy have been streamlined, moving from monthly paperwork to a one-time enrollment agreement.Common Pitfalls and How to Avoid Them
Even with the new updates, the system can still glitch. Pharmacy system errors are a common reason for delays. If your pharmacy says they "can't see you in the system," don't panic. Usually, it's a synchronization error between the pharmacy's software and the iPLEDGE database. Another common issue is the timing of the pregnancy test. Even with home tests, the timing must be precise. If you test too early, your doctor cannot "authorize" the prescription in the portal, and the pharmacy cannot fill it. Always coordinate your test date with your doctor's office a few days before your prescription is due to refill. If you run into a wall, there is a 24/7 helpline at 1-866-495-0654, though be prepared for wait times during peak hours.
Comparing iPLEDGE to Older Systems
Before iPLEDGE, there was a program called SMART (System to Manage Accutane-Related Teratogenicity). The main difference was that SMART was brand-specific. If you switched from one brand of isotretinoin to another, you had to register all over again. iPLEDGE fixed this by creating a single, centralized platform for all manufacturers, including Mylan and Teva. However, controversy still exists. Some studies, including a 2011 report in the Journal of the American Academy of Dermatology, suggest that iPLEDGE hasn't actually been more effective than SMART at reducing fetal exposure. Critics argue that the administrative burden-where some doctors spend up to 7 hours a week just managing the software-creates barriers to care without providing a proportional increase in safety. Despite this, the FDA maintains that the risk of the drug is so extreme that the high level of control is the only acceptable way to manage it.Can I start isotretinoin if I am currently pregnant?
Absolutely not. Isotretinoin causes severe birth defects. The iPLEDGE program strictly forbids any female patient from starting therapy if they are pregnant. You must provide two negative pregnancy tests before the first dose is dispensed.
What happens if I miss my monthly pregnancy test?
If you miss the window for your monthly test, your prescriber cannot authorize your next refill in the iPLEDGE system. This will result in a delay in receiving your medication. You will need to complete the test and have your doctor update the system before the pharmacy can release the drug.
Do men have to participate in iPLEDGE?
Yes. While the pregnancy testing and contraception requirements only apply to those capable of pregnancy, all patients regardless of gender must be registered in the iPLEDGE system and acknowledge the risks of the medication.
Are home pregnancy tests always allowed?
Under the November 2023 updates, home pregnancy testing is permitted, but it is up to your individual prescriber to implement a verification protocol. Some doctors may still require a clinic-based test for the very first dose of the treatment cycle.
How long does the treatment typically last?
Most patients stay on isotretinoin for 4 to 5 months. Because the REMS requirements are monthly, you can expect to go through the testing and authorization process about 4 or 5 times during your course of treatment.





Majestic Blue Band April 26, 2026
It is absolutely laughable that we are expected to just trust a "centralized web-based system" managed by the government and big pharma when we all know these databases are just tools for mass surveillance and control of our bodily autonomy, and honestly, the fact that they claim these birth defects are the only reason for such an invasive system is probably a cover for some larger experimental social engineering project designed to see how much bureaucracy the average citizen will put up with before they snap, especially since the 2011 report already proved the system is useless, which clearly means the whole thing is a giant scam to make dermatologists feel important while the FDA collects data on our movements and health statuses in real-time.
Edwin Perez April 28, 2026
just another way for the feds to track every move you make while pretending to care about safety. typical
Elle Torres Sanz April 30, 2026
It's really important to remember that different healthcare systems around the world handle this differently, but for those in the US, following these steps is the safest bet to get the results you want. Let's just try to support each other through the paperwork struggle.
Andre Ojakäär April 30, 2026
absolute madness that we still use this portal in 2024 it is literally a prehistoric piece of software that crashes if you even look at it wrong i had to call my doctor three times just to fix one checkbox
Vijay AGarwal May 1, 2026
OH MY GOD the stress of that synchronization error is a nightmare! I have seen patients literally break down in tears at the pharmacy counter because the system says they aren't authorized even though they did the test! IT IS ABSOLUTE CHAOS!
Sharyl Foster May 2, 2026
Actually, the home tests are only a 'game-changer' if your doctor isn't a dinosaur who still insists on clinic visits. Most of them still make you come in because they like the control, so the 'update' is barely an update at all.
Jon Moss May 3, 2026
yeah that sounds about right with most derm offices
Michael Chukwuma May 5, 2026
I can totally see why that would be frustrating. It's a lot to manage on top of dealing with severe acne, but it's good to know there are ways to make it easier now.
suresh kumar May 5, 2026
This whole thing is a circus of red tape! I know a guy who almost threw his laptop across the room trying to sign the digital agreement because the website is a clunky piece of garbage that belongs in a museum of failure!
Eric Mwiti May 6, 2026
Oh sure, because a 24/7 helpline with massive wait times is exactly what every patient needs in a crisis. Truly a pinnacle of government efficiency.
Karyn Tindall May 6, 2026
I am absolutely horrified by the description of the birth defects! It is just terrifying to think that one mistake could lead to such a tragedy! The strictness is completely justified!
Beena Garud May 6, 2026
One must contemplate the intersection of medical necessity and the ethical imperative of fetal protection. The administrative burden is a small price for the preservation of life.