Drug shortages in the U.S. aren’t just inconveniences-they’re life-or-death emergencies. In 2024, over 300 drugs were in short supply, including antibiotics, cancer treatments, and anesthesia. Hospitals scrambled. Patients delayed care. Pharmacists spent hours tracking down substitutes. The federal government didn’t sit idle. By 2025, it launched its most aggressive effort yet: the Strategic Active Pharmaceutical Ingredients Reserve (SAPIR). But is it working? And what’s really behind the shortages?
What’s Causing the Shortages?
It’s not one problem. It’s a broken system. About 80% of the raw ingredients for U.S. drugs come from China and India. When a factory there shuts down for inspection, or a natural disaster hits, or a company decides it’s not profitable to make low-cost generic drugs, the ripple effect hits American hospitals hard. Sterile injectables-like saline, insulin, and chemotherapy drugs-are especially vulnerable. Just five facilities produce 78% of these critical medicines. One glitch, and thousands of patients are at risk.The FDA says 98 drugs were still in shortage at the end of 2024. But other groups report higher numbers. Why? Because they count different things. The FDA tracks only active, confirmed shortages. Others include near-misses and delays. Either way, the impact is real. Hospitals now spend an average of $1.2 million a year just managing shortages. Pharmacists report working 10+ hours a week tracking down pills. Some have had to compound cancer drugs from raw powder because nothing else was available.
The SAPIR Initiative: Stockpiling Raw Ingredients
In August 2025, President Trump signed Executive Order 14178, expanding the SAPIR program. This isn’t about hoarding finished drugs. It’s about storing the raw chemicals-Active Pharmaceutical Ingredients (APIs)-that go into them. Why? Because APIs cost 40-60% less than finished pills, last 3-5 years longer in storage, and are easier to ship. The goal: stockpile APIs for 26 essential drugs, including antibiotics, anesthetics, and oncology treatments.The idea makes sense on paper. If a factory in China shuts down, the U.S. can quickly turn its stored APIs into medicine. But there’s a catch. The 26 drugs on the list cover only 4% of all shortage-prone medications. Meanwhile, oncology drugs-which made up 31% of all shortages in 2024-are barely represented. Experts like Dr. Peter Pitts warn this is like building a fire alarm for one room while the whole house burns down.
FDA’s Role: Speed, Not Solutions
The FDA doesn’t just sit back. It’s actively working with manufacturers to fix shortages. In 85% of cases, the agency helps by speeding up inspections, approving temporary imports, or allowing alternative manufacturing methods. The 2018-2020 saline shortage was resolved this way-after 90% of hospitals ran out, the FDA fast-tracked imports from Europe and helped a U.S. plant ramp up production.But the FDA’s biggest tool-mandatory shortage reporting-isn’t working. By law, manufacturers must report potential shortages six months in advance. Yet only 58% comply. Small companies? Only 18% report. That means the FDA often finds out about a shortage after it’s already started. In response, they launched the Enhanced Shortage Monitoring System in November 2025. It uses AI to predict shortages 90 days ahead by analyzing shipping data, factory output, and hospital purchase patterns. So far, it’s 82% accurate. That’s promising. But without better reporting from drugmakers, it’s still playing catch-up.

The Bigger Picture: Why Manufacturing Won’t Come Home
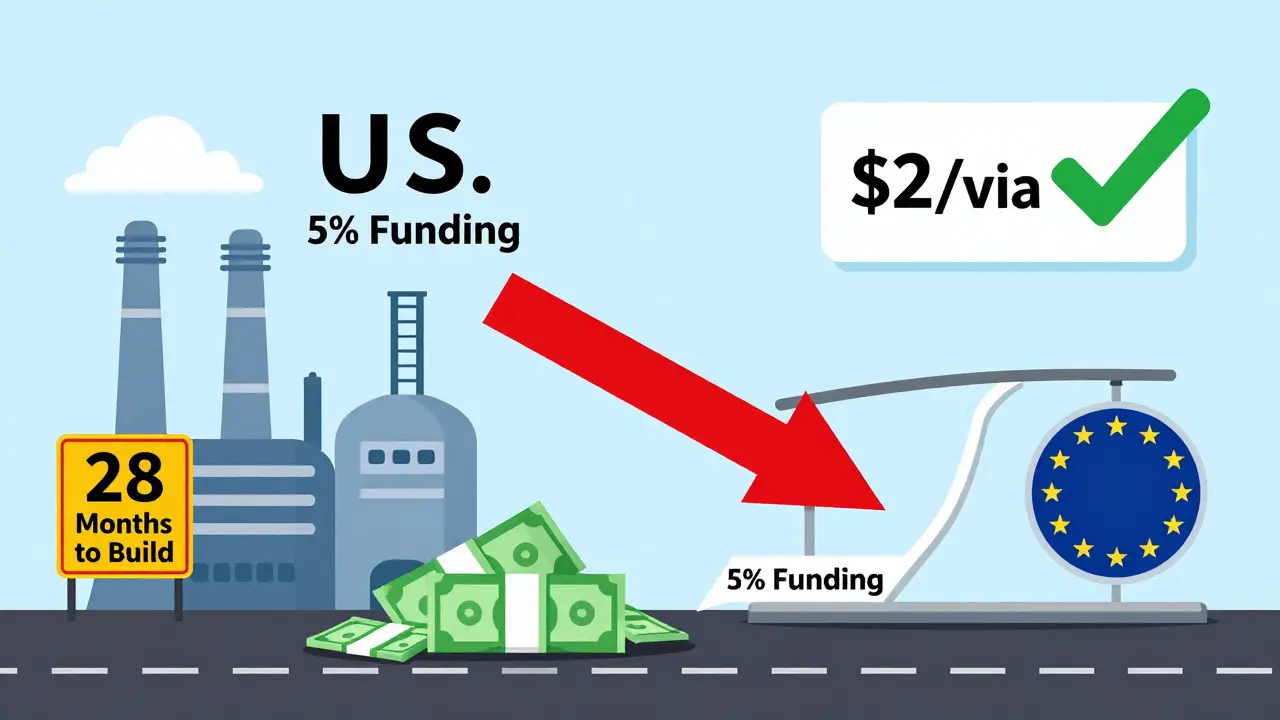
You’d think the answer is to bring drug production back to the U.S. But it’s not that simple. Building a new API facility takes 28-36 months here, compared to 18-24 in the EU. Regulatory hurdles are higher. And profit margins? Thin. A single vial of generic antibiotic might sell for $2. The cost to produce it? $1.75. Who’s going to invest millions to build a plant for that?The federal government tried to fix this with funding. In September 2025, the Department of Commerce announced $285 million in CHIPS Act money for domestic pharmaceutical plants. Sounds good. But industry analysts say it covers less than 5% of what’s needed. Meanwhile, NIH’s drug development budget dropped 18% from 2024 to 2025. That’s not a signal to build-it’s a signal to wait.
What’s Missing: Economic Incentives
The real problem isn’t just supply chains. It’s economics. Companies make money on blockbuster drugs. Not on cheap generics. So they stop making them. The EU solved this by requiring member states to maintain minimum stockpiles and pay manufacturers a guaranteed price. The U.S. hasn’t done that. Instead, it’s relying on voluntary reporting and emergency stockpiles.There’s a bill in Congress-H.R.5316-that would create Medicare payments to hospitals that maintain backup supply chains during shortages. The American Hospital Association supports it. But critics say it’s a band-aid. If hospitals are paying extra to stockpile drugs, why not pay drugmakers to make them? The FDA’s own data shows that the most effective solution so far is the Early Notification Pilot Program, which cut shortage durations by 28%. That program worked because it required manufacturers to report early-and enforced it.
Where the System Is Failing
The Government Accountability Office found that HHS has no clear way to measure whether its shortage programs are working. Only 35% of ASPR’s recommended actions have been adopted across agencies. States? Only 28 out of 50 have set up the supply chain mapping tools required by the 2025 Action Plan. Rural hospitals, already understaffed, say it takes 3-6 months just to get the software running.And then there’s enforcement. Between 2020 and 2024, the FDA issued just 17 warning letters for failure to report shortages. The EU issued 142 under similar rules. That’s not oversight. That’s neglect.
What Patients Are Experiencing
Real people are paying the price. In September 2025, Patients for Affordable Drugs reported that 29% of Americans skipped doses because their drug wasn’t available. Cancer patients were hit hardest-68% reported treatment delays or substitutions. On Reddit, pharmacists described switching between five different manufacturers for the same drug in one week. One wrote: "I had to compound cisplatin from raw chemicals. We had no choice."These aren’t rare cases. The American Society of Health-System Pharmacists found 41% of pharmacists had experienced near-miss errors directly tied to shortage-related substitutions. That means someone almost got the wrong dose, or the wrong drug, because the original wasn’t in stock.
Is There a Better Way?
The EU’s model is working. Mandatory stockpiling. Centralized tracking. Guaranteed pricing. Their shortages dropped 37% between 2022 and 2024. The U.S. could do the same. But instead, it’s doubling down on stockpiling APIs while cutting funding for prevention, weakening reporting rules, and ignoring the economic realities that make drug production unprofitable.Until the government starts paying manufacturers fairly to produce essential medicines-and enforces transparency-shortages will keep coming. Stockpiling raw ingredients helps in a crisis. But it doesn’t fix the system. And right now, the system is failing patients every day.
What is the Strategic Active Pharmaceutical Ingredients Reserve (SAPIR)?
SAPIR is a federal program launched in 2020 and expanded in August 2025 to stockpile the raw chemical ingredients (APIs) used to make 26 critical drugs, including antibiotics, anesthetics, and cancer treatments. The goal is to prevent shortages by having these ingredients on hand so they can be quickly turned into finished medications during emergencies. APIs are cheaper and last longer than finished drugs, making them more practical to store. However, the program currently covers only 4% of drugs that commonly go into shortage.
Why are so many drug shortages happening in the U.S.?
Most drug shortages stem from a fragile supply chain. About 80% of active pharmaceutical ingredients come from just two countries-China and India. Production is also concentrated: 78% of sterile injectables are made in only five facilities. When one factory has a problem-whether from inspection delays, equipment failure, or economic pressure to stop making low-margin drugs-the entire system feels it. Weak reporting rules and lack of financial incentives for manufacturers to produce generics make the problem worse.
How is the FDA responding to drug shortages?
The FDA works directly with manufacturers to resolve shortages by expediting inspections, approving temporary imports, and allowing alternative production methods. It also runs the Enhanced Shortage Monitoring System, an AI tool that predicts shortages 90 days in advance with 82% accuracy by analyzing shipping data, manufacturing output, and hospital purchasing trends. However, enforcement of mandatory shortage reporting remains weak, with only 58% of manufacturers complying, and small companies reporting at even lower rates.
Why hasn’t drug manufacturing returned to the U.S.?
Building a new drug manufacturing facility in the U.S. takes 28-36 months-longer than in the EU or Asia. Regulatory hurdles are stricter, and the profit margins on essential generic drugs are razor-thin. A single vial of generic antibiotic might cost $1.75 to make and sell for $2. There’s little financial incentive for companies to invest millions in domestic plants. Federal funding efforts, like the $285 million CHIPS Act allocation, cover less than 5% of what’s needed to make a real difference.
What’s the most effective solution to drug shortages?
The most proven solution is mandatory early reporting and financial incentives for manufacturers. The FDA’s Early Notification Pilot Program reduced shortage durations by 28% because it required companies to report potential problems months in advance. The EU’s system-mandatory stockpiling, centralized tracking, and guaranteed pricing-cut shortages by 37% over two years. In the U.S., legislation like H.R.5316 proposes Medicare payments to hospitals for maintaining backup supplies, but experts argue that paying manufacturers to produce essential drugs reliably is the only long-term fix.





Kandace Bennett March 15, 2026
Okay but let’s be real-SAPIR is just a PR stunt with a fancy acronym. 🤦♀️ We’re stockpiling 26 drugs while 300+ are in crisis? That’s like buying one umbrella for a hurricane. The real issue? Pharma companies stopped making generics because they’re dirt cheap. Why would you invest millions for $2 vials? 🤔 The EU pays manufacturers to produce. We just yell at China. 🇺🇸❌
Tim Schulz March 15, 2026
Oh wow. A federal program that *actually* does something? 🤯 I’m shocked. Let me grab my popcorn while the FDA plays whack-a-mole with 98 shortages and calls it a ‘solution.’ Meanwhile, pharmacists are compounding chemo from powder because the system’s been on life support since 2010. 😅
Jinesh Jain March 16, 2026
Interesting perspective. In India, we see how API production works-both the efficiency and the fragility. Many small manufacturers here supply global markets, but quality control is uneven. A single factory shutdown in China affects us too. Maybe cooperation-not just stockpiling-is the real answer.
douglas martinez March 18, 2026
The structural challenges in pharmaceutical manufacturing are profound and require systemic reform. While the Strategic Active Pharmaceutical Ingredients Reserve represents a tactical response, it does not address the underlying economic disincentives for domestic production. A comprehensive policy framework must include price stabilization, regulatory streamlining, and long-term investment incentives.
Sabrina Sanches March 18, 2026
I just want my insulin to be available. That’s it. No drama. No politics. Just medicine. 🥺
Shruti Chaturvedi March 19, 2026
From India to the U.S.-we’re all connected in this supply chain. It’s not about blame. It’s about building trust. Maybe if we stopped seeing each other as enemies and started seeing each other as partners, we could fix this faster. Simple idea. Hard to do. But worth trying.
Katherine Rodriguez March 20, 2026
China’s the problem. Always is. They control the APIs. They hold the U.S. hostage. We need tariffs. We need bans. We need to cut them off. Why are we letting them run our medicine? This isn’t globalization-it’s surrender.
Devin Ersoy March 21, 2026
Let’s be gloriously unapologetic here: SAPIR is a Band-Aid on a severed artery. We’re not fixing the system-we’re just slapping glitter on the corpse. The FDA’s AI predictor? Cute. But if manufacturers aren’t forced to report, it’s just a fancy spreadsheet with delusions of grandeur. 🎩✨
Scott Smith March 23, 2026
There’s a lot here worth considering. I appreciate the data. But I also think we need to hear more from the pharmacists on the front lines. They’re not just logistics coordinators-they’re patient advocates. Their stories matter more than any policy document.
Sally Lloyd March 24, 2026
Did you know the FDA’s AI system was trained on data from a single pharmaceutical conglomerate? Coincidence? Or is this all just a controlled narrative? Who owns the servers? Who funds the algorithm? The same companies that profit from shortages? 🤔
Emma Deasy March 25, 2026
It is, without question, a national tragedy-this systematic erosion of medical security. Each day, a patient waits. Each week, a life hangs in the balance. And still, we debate metrics and budgets and timelines. Where is the moral urgency? Where is the collective outrage? We are not just managing shortages-we are burying lives with bureaucracy.
tamilan Nadar March 27, 2026
As someone who works in pharma logistics in India, I can say this: the U.S. isn’t alone. We face the same issues-low margins, poor reporting, no incentives. But we also have innovation. Small labs here are making APIs cheaper and faster. Maybe the answer isn’t stockpiling-it’s decentralizing production.
Adam M March 28, 2026
SAPIR’s useless. 4% coverage. Pathetic.
Rosemary Chude-Sokei March 28, 2026
I appreciate the depth of this analysis. It’s clear that the issue is multi-layered and requires collaboration across federal agencies, industry stakeholders, and international partners. While SAPIR is a step in the right direction, true progress will require sustained funding, enforceable reporting mandates, and a shift in how we value essential medicines-not as commodities, but as public goods.
Noluthando Devour Mamabolo March 30, 2026
From a supply chain analytics standpoint, the SAPIR initiative exhibits a high degree of strategic alignment with risk mitigation frameworks. However, the absence of dynamic inventory replenishment algorithms and API traceability via blockchain integration renders the system vulnerable to latency and opacity. We need real-time, federated data ecosystems-not static stockpiles.