Portal vein thrombosis (PVT) isn’t something most people hear about until it’s already affecting their liver or digestive system. But when it happens, it can turn a manageable liver condition into a life-threatening one. The portal vein carries blood from your intestines to your liver. When a clot blocks it, blood backs up. That raises pressure in the portal system, strains the liver, and can starve parts of the intestine of oxygen. The good news? When caught early and treated right, most people recover well. The bad news? Many cases are missed because symptoms are vague - bloating, mild abdominal pain, or just feeling off. By the time someone gets scanned, the clot might have been there for months.
What exactly is portal vein thrombosis?
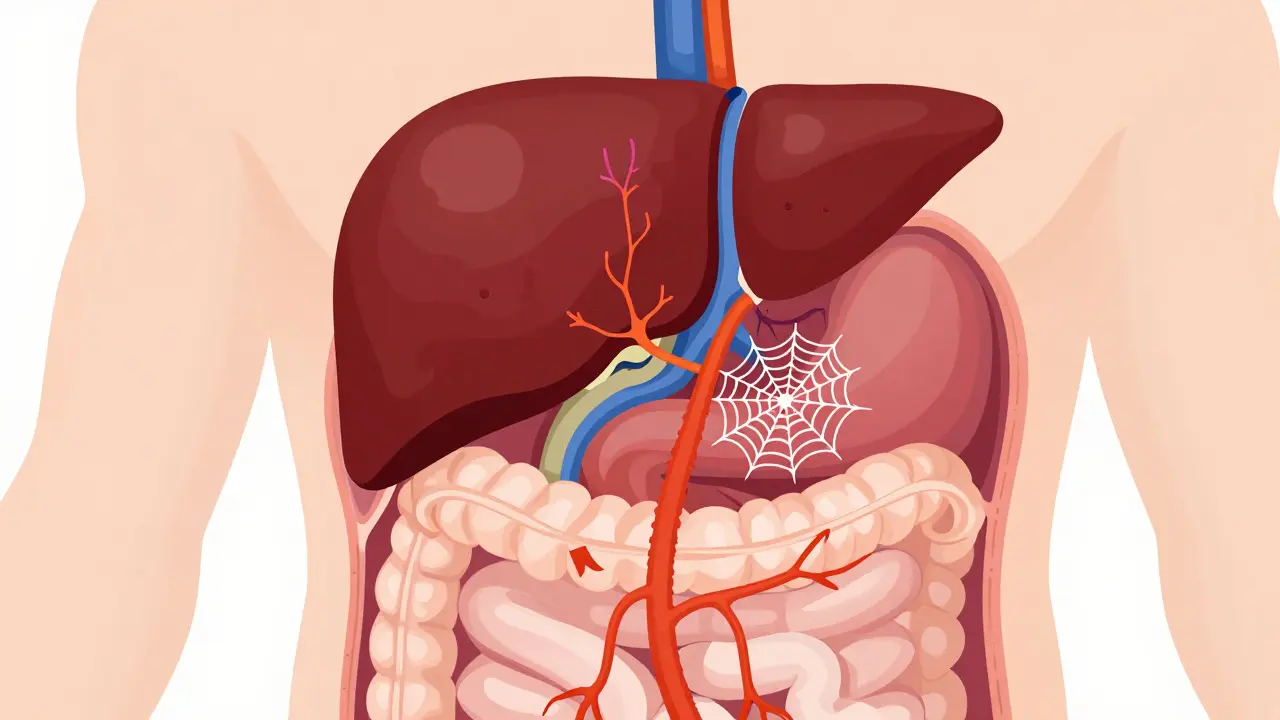
Portal vein thrombosis means a blood clot has formed inside the main vein that brings blood from your stomach, intestines, and spleen to your liver. It can be partial or complete. Sometimes it’s just a small clot; other times, the whole vein is blocked. In chronic cases, the body tries to compensate by growing new tiny blood vessels around the blocked area - a process called cavernous transformation. This sounds helpful, but it doesn’t restore normal blood flow. It just makes things more complicated.
PVT isn’t one-size-fits-all. It’s either acute - coming on fast, usually within two weeks - or chronic, where the clot has been there for more than six weeks. Acute PVT is more treatable. If you act fast, the clot can dissolve. Chronic PVT is harder to reverse. That’s why timing matters more than almost anything else.
How do doctors diagnose it?
Ultrasound is the first step. It’s cheap, quick, and doesn’t use radiation. A skilled technician can see if the portal vein is narrowed, filled with clot, or completely gone. Doppler ultrasound checks blood flow - if there’s no flow where there should be, that’s a red flag. The test catches PVT in 89% to 94% of cases. If the ultrasound is unclear, a CT scan or MRI with contrast is next. These show the clot in detail and whether it’s spread into the branches inside the liver.
Doctors also look for signs of liver damage. They check liver function tests, but those alone won’t confirm PVT. What they really need to know is whether you have cirrhosis, and if so, how advanced it is. That’s measured using the Child-Pugh score or MELD score. These scores help decide if anticoagulation is safe. A patient with Child-Pugh C cirrhosis - meaning severe liver failure - has a much higher risk of bleeding. Giving them blood thinners could be dangerous.
Another key test: endoscopy. If you have cirrhosis, you’re likely to have swollen veins in your esophagus or stomach - varices. These can bleed easily. If you have PVT and varices, you need to treat the varices before starting anticoagulation. Otherwise, the clot treatment could trigger a life-threatening bleed.
Why anticoagulation is the standard - and when it’s not
For years, doctors were scared to give blood thinners to people with liver disease. They thought it would cause bleeding. But over the last decade, data changed everything. Studies now show that for most people with acute PVT - even those with cirrhosis - anticoagulation improves survival and helps the clot dissolve. The 5-year survival rate jumps to 85% with treatment, compared to under 50% without it.
Anticoagulation isn’t about preventing new clots. It’s about dissolving the existing one. It stops the clot from growing, helps the body break it down, and prevents complications like intestinal ischemia - where parts of the bowel don’t get enough blood and start dying.
But it’s not for everyone. You shouldn’t get anticoagulation if:
- You had a variceal bleed in the last 30 days
- You have uncontrolled ascites (fluid in the belly)
- Your liver function is severely damaged (Child-Pugh C)
For cirrhotic patients with Child-Pugh A or B, anticoagulation is now recommended - as long as varices are treated first. In fact, studies show that treating varices with band ligation before starting anticoagulation cuts major bleeding risk from 15% down to 4%.
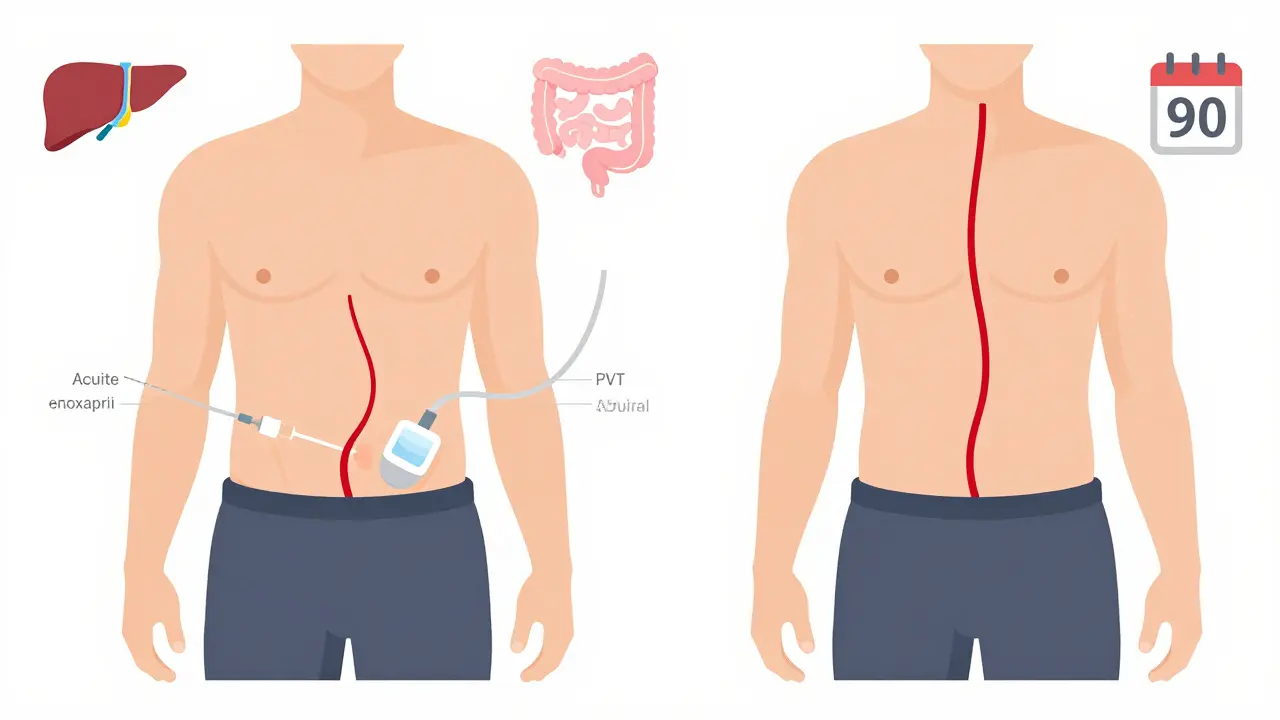
Which blood thinners work best?
There are three main types: low molecular weight heparin (LMWH), vitamin K antagonists (like warfarin), and direct oral anticoagulants (DOACs).
LMWH - like enoxaparin - is often used first. It’s given by injection, usually once or twice a day. It’s predictable, doesn’t need frequent blood tests, and works well in cirrhotic patients. For non-cirrhotic patients, DOACs are now preferred. Rivaroxaban, apixaban, and dabigatran have shown better recanalization rates than warfarin. One study found 65% to 75% of non-cirrhotic patients on DOACs had complete clot resolution, compared to just 40-50% on warfarin.
DOACs are easier. You take one pill a day. No weekly INR checks. But they’re not for everyone. If your kidneys are failing or you have Child-Pugh C cirrhosis, DOACs can be risky. The FDA has black box warnings for severe liver disease. That’s why LMWH is still the go-to for many cirrhotic patients.
Here’s what works best:
| Drug Class | Typical Dose | Best For | Recanalization Rate | Bleeding Risk |
|---|---|---|---|---|
| LMWH (enoxaparin) | 1 mg/kg twice daily or 1.5 mg/kg once daily | Cirrhotic patients (Child-Pugh A/B) | 55-65% | 5-12% |
| Rivaroxaban (DOAC) | 20 mg daily | Non-cirrhotic patients | 65% | 2-5% |
| Apixaban (DOAC) | 5 mg twice daily | Non-cirrhotic patients | 65% | 2-5% |
| Warfarin (VKA) | Target INR 2.0-3.0 | When DOACs/LMWH aren’t available | 40-50% | 4-8% |
Duration of treatment also matters. If the clot was caused by something temporary - like recent surgery or an infection - six months of anticoagulation is usually enough. But if you have a genetic clotting disorder (like Factor V Leiden), or cancer, lifelong treatment is needed. About 25-30% of non-cirrhotic PVT patients have an underlying clotting condition.
What if anticoagulation doesn’t work?
Some clots don’t dissolve. If the clot is still blocking the portal vein after six months of anticoagulation, or if the patient develops worsening portal hypertension, other options come in.
Transjugular Intrahepatic Portosystemic Shunt (TIPS) is one. It creates a tunnel inside the liver to bypass the blocked vein. Success rates are 70-80%, but it can cause confusion or drowsiness (hepatic encephalopathy) in 15-25% of cases. It’s not a first-line fix - it’s used when anticoagulation fails.
Percutaneous thrombectomy is another option. A catheter is threaded in to physically break up the clot. It works well - 60-75% immediate success - but only a few specialized centers do it. It’s expensive and risky.
For patients waiting for a liver transplant, anticoagulation is critical. Studies show those on anticoagulants have an 85% one-year survival rate after transplant - compared to 65% without. PVT used to be a reason to remove someone from the transplant list. Now, it’s often treated and reversed.
Real-world challenges and what works
At Mayo Clinic, 78% of patients treated within 30 days of diagnosis saw clot resolution. But only 42% of those treated after 90 days did. Delayed diagnosis kills outcomes.
UCSF’s transplant program found that anticoagulation cut the number of patients being denied transplant listings due to PVT from 22% to 8%. That’s huge.
But it’s not easy. Many community doctors still hesitate. Only 35% of general gastroenterologists feel confident managing PVT anticoagulation. That’s why coordination matters. The best outcomes happen when hepatologists, radiologists, and endoscopists work together. One hospital reduced complications by 35% just by setting up regular team meetings.
Another key insight: platelet count matters. If your platelets are below 50,000/μL, giving anticoagulants can be risky. Some centers give a platelet transfusion to raise counts above 30,000/μL before starting treatment. It’s safe and effective.
The future: DOACs, new drugs, and precision care
The landscape is shifting fast. In 2024, the AASLD updated its guidelines to allow DOACs in Child-Pugh B7 patients - a group previously excluded. The CAVES trial showed DOACs were just as effective as LMWH in this group.
Andexanet alfa, approved in 2023, is a game-changer. It’s a reversal agent for rivaroxaban and apixaban. If a patient on DOACs starts bleeding badly, this drug can stop it quickly. That’s made doctors more comfortable prescribing DOACs.
By 2025, experts predict 75% of non-cirrhotic PVT cases will be treated with DOACs. Even in compensated cirrhosis, use will rise. New drugs like abelacimab are in trials. But for now, anticoagulation remains the backbone of treatment.
One thing’s clear: if you have PVT and aren’t on anticoagulation - and you don’t have active bleeding or severe liver failure - you’re likely missing out on a treatment that could save your liver, your intestines, and maybe your life.
Can portal vein thrombosis be reversed?
Yes, especially if caught early. With anticoagulation started within six months, 65-75% of patients see complete or partial clot resolution. The longer you wait, the lower the chance. Chronic PVT is harder to reverse, but treatment still prevents complications like intestinal ischemia and worsening liver damage.
Is anticoagulation safe for people with cirrhosis?
It can be, but only if liver function isn’t too damaged. For Child-Pugh A or B cirrhosis, anticoagulation is recommended - especially if varices have been treated with band ligation. For Child-Pugh C, it’s usually avoided due to high bleeding risk. LMWH is preferred over DOACs in cirrhotic patients because it’s more predictable and doesn’t rely on liver metabolism.
Why do some doctors avoid giving blood thinners for PVT?
Historically, doctors feared bleeding, especially in cirrhotic patients with varices. But newer data shows that the risk of not treating - intestinal ischemia, clot extension, transplant rejection - is far greater. The key is proper patient selection: treat varices first, avoid anticoagulation in active bleeding or severe liver failure, and use the right drug for the patient’s liver function.
Do I need lifelong anticoagulation after PVT?
Not always. If the clot was caused by a temporary trigger - like surgery or infection - six months is usually enough. But if you have a genetic clotting disorder (like Factor V Leiden) or cancer, lifelong treatment is needed. About one in four non-cirrhotic patients has an underlying clotting condition that requires long-term therapy.
Can I take DOACs if I have liver disease?
DOACs like rivaroxaban and apixaban are safe for patients with mild to moderate liver disease (Child-Pugh A or B), especially if they’re non-cirrhotic. They’re now preferred in these groups because they’re easier to manage and more effective than warfarin. But they’re not recommended for severe liver failure (Child-Pugh C) due to increased bleeding risk. Always check kidney function too - DOACs are cleared by the kidneys.



Allison Priole March 22, 2026
i’ve been following this since my cousin got diagnosed last year. honestly? the fact that we can now treat this with pills instead of injections is wild. my aunt was on enoxaparin for 6 months and now her scan looks clean. no more bloating, no more pain. just normal life. i know people think liver stuff is all doom and gloom but this is proof that medicine is actually getting better. also, DOACs are a game changer for non-cirrhotic folks. why make someone inject themselves daily when a pill works better?
ps: if you’re scared of bleeding, talk to your doc about variceal banding first. it’s not scary. it’s like a tiny rubber band for your veins. weird but life-saving.
Casey Tenney March 24, 2026
If you’re not on anticoagulation and you have PVT, you’re playing Russian roulette with your liver.
Bryan Woody March 25, 2026
let me guess - some doc out there is still telling patients to avoid blood thinners because ‘liver = fragile’ like it’s 1998. newsflash: we have data now. not opinions. not fear. data.
the studies are in. 85% survival with anticoagulation vs 50% without. that’s not a suggestion. that’s a mandate. and yet, 35% of gastro docs still don’t feel confident managing this? that’s not incompetence. that’s negligence wrapped in a white coat.
doac’s in child-pugh b7? yes. and the caves trial proved it. if your liver can handle a glass of wine, it can handle rivaroxaban. stop treating cirrhosis like a death sentence and start treating it like a chronic condition. we’ve got reversal agents now. we’ve got banding. we’ve got timing. we’ve got science. why are we still whispering about this?
Timothy Olcott March 25, 2026
this is why america is falling behind 🇺🇸
we have the best science on earth but half the docs are still scared of pills. in india they use DOACs for everything. even with cirrhosis. no big deal. here? we make it complicated.
also platelet transfusion before anticoag? that’s genius. why didn’t we think of that 20 years ago? 🤦♂️
Johny Prayogi March 27, 2026
this is the kind of info that should be in med school textbooks. not buried in a 10k word article. i’m so glad we’re finally seeing real progress. the shift from ‘don’t touch anticoag’ to ‘here’s the exact protocol’ is huge. and the fact that transplant eligibility improved from 22% to 8% denial? that’s not just stats. that’s people getting second chances. thank you to every researcher who pushed this forward. you’re saving lives without even knowing their names.
Nishan Basnet March 27, 2026
what struck me most is how much of this is about timing, not just drugs. the difference between 30 days and 90 days of delay isn’t just statistical - it’s existential. one person gets to go back to work. another gets a shunt. another gets denied transplant. it’s not about the clot. it’s about the clock. and we’re still too slow to react. if we had a national PVT alert system - like stroke or heart attack - outcomes would change overnight. this isn’t rare. it’s just ignored.
Sandy Wells March 29, 2026
I find it concerning that so many physicians are still hesitant to prescribe anticoagulation in cirrhotic patients despite overwhelming evidence. The hesitation is not evidence-based. It is rooted in outdated protocols and fear of litigation. One must ask: is the fear of bleeding greater than the certainty of ischemia? The answer is clear. The data is not ambiguous. The guidelines are not opinion. They are science.
Jackie Tucker March 30, 2026
so let me get this straight - we’re using blood thinners to fix a clot that was caused by... liver disease? which is caused by... alcohol? or obesity? or bad genes?
so we’re medicating the symptom of a symptom of a symptom?
what if we just... stopped making people eat processed food? or drink? or sit on their butts?
instead we give them rivaroxaban and call it ‘precision medicine’. how poetic. how tragic. how… american.
matthew runcie March 31, 2026
glad to see this getting attention. i’ve seen too many patients wait too long because their doc said ‘it’s probably nothing’. the ultrasound is cheap. the risk of waiting is huge. just get it checked. no one’s asking you to start a drug regimen on day one. just get the scan. it takes 15 minutes. your liver will thank you.
Nicole James April 1, 2026
this is all a distraction... the real cause of PVT? 5G towers. they interfere with portal vein blood flow. they’ve been suppressing the immune response for decades. you think the FDA cares? they’re paid off by big pharma. DOACs? they’re just a way to keep you dependent. the truth? it’s the EMFs. the government won’t admit it. but i’ve read the studies. they’re buried. just like the truth.
ask yourself: why are they pushing anticoagulation? because they want you to need more pills. more scans. more money. it’s not about healing. it’s about control.
Solomon Kindie April 1, 2026
the fact that we’re even debating this is insane. we have a clot in a major vein leading to the liver and we’re talking about which pill to give? we should be doing emergency thrombectomy on day one. not waiting for ultrasounds. not waiting for variceal banding. not waiting for liver scores. we’re treating this like a chronic condition when it’s an emergency. we need a pvt code blue. like stroke. like mi. not ‘let’s schedule an appointment next tuesday’
Natali Shevchenko April 3, 2026
i’ve been thinking about this a lot lately. the body’s not broken when it forms a clot. it’s trying to protect itself. maybe the clot isn’t the enemy. maybe it’s the signal. the liver’s saying ‘hey, something’s off’. we’re so obsessed with dissolving it that we forget to ask why it formed in the first place.
is it inflammation? is it poor diet? is it silent celiac? is it a hidden cancer?
anticoagulation fixes the symptom. but what if the real cure is in the quiet space between the scan and the pill? what if healing starts before the drug? what if we stopped treating clots and started listening to bodies?
Chris Dwyer April 3, 2026
you guys are overcomplicating this. if you have pvt and you’re not bleeding and your liver isn’t falling apart - just start the anticoagulation. no need for a philosophy degree. no need to wait for 3 specialists to agree.
lmwh for cirrhotic. doac for non-cirrhotic. band the varices first. done.
the data’s clear. the guidelines are simple. the only thing holding people back is fear. and fear doesn’t save lives. action does. go get your scan. talk to your doc. take the pill. your future self will high-five you.
Desiree LaPointe April 5, 2026
how convenient that the same pharmaceutical companies that profit from DOACs are also funding the ‘guidelines’. how poetic that we’ve turned a life-threatening clot into a $12,000/year subscription service.
and yet - here we are. celebrating ‘progress’ while ignoring the elephant in the room: if we stopped promoting these drugs as first-line, and started promoting lifestyle, nutrition, and early screening - would we even need anticoagulation?
the answer is yes. we’d need less. much less.
but then… who profits?