Quick Takeaways
- Generics use the same active ingredients and work the same way in the body as brand names.
- You can expect massive savings, often 80% to 85% less than the brand version.
- Differences in color, shape, and inactive fillers are normal and required by law.
- Certain "narrow therapeutic index" drugs require closer monitoring when switching.
- Always keep a simple log of your symptoms for two weeks after a switch.
What Exactly Is a Generic Medication?
Think of a generic drug as a store-brand version of a name-brand product. It is a medication created to be the same as an already marketed brand-name drug in dosage form, strength, route of administration, quality, performance characteristics, and intended use. When a pharmaceutical company first creates a drug, they get a patent that lasts about 20 years. Once that patent expires, other companies can apply to the FDA to make their own version.
To get approval, generic makers don't have to redo the massive clinical trials the original company did. Instead, they prove bioequivalence. This means the drug enters the bloodstream at the same rate and to the same extent as the brand name. Current regulations require that the generic's performance falls within a strict 80% to 125% confidence interval of the brand's pharmacokinetic parameters. In reality, the gap is even smaller; data from Harvard Medical School shows the actual observed variation is usually only around 4.1%.
The Price Gap: Why Your Wallet Will Thank You
The most immediate thing you'll notice after switching is the cost. Brand-name drugs are expensive because the original company had to recoup the billions spent on research and development. Generics don't have those overhead costs, and they compete with each other, which drives prices down.
| Feature | Brand-Name Medication | Generic Equivalent |
|---|---|---|
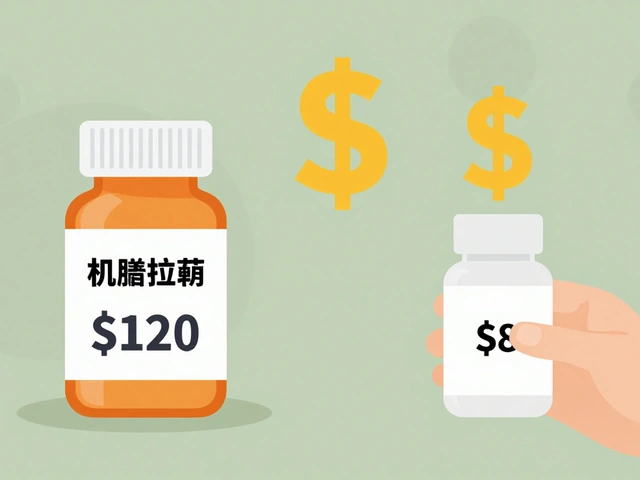
| Average Cost per Prescription | $624 | $128 |
| Typical Price Reduction | Reference Point | 80% to 85% Lower |
| Annual Medicare Savings | - | ~$1,268 (Average) |
For many, this isn't just about extra spending money-it's about whether they can afford the medicine at all. Consider a patient taking a cholesterol medication; paying $350 a month might lead to skipping doses, while a $15 generic ensures they take every single pill. This higher adherence rate actually leads to better health outcomes than the "premium" brand drug that stays in the pharmacy bottle because it's too expensive.
What Might Feel Different (And Why)
If the active ingredient is identical, why does the pill look different? It comes down to trademark laws. The FDA requires generics to look different from the original to avoid trademark infringement. This means the shape, size, and color will almost always vary.
Then there are the inactive ingredients. These are the fillers, binders, and dyes that hold the pill together. While the active drug is the same, the "glue" isn't. For 99% of people, this makes no difference. However, if you have a severe allergy to a specific dye or a sensitivity to lactose or gluten (which can sometimes be used as fillers), you might react to a generic even if you tolerated the brand. This is why checking the Patient Package Insert (PPI) is a smart move.

When a Switch Requires Extra Caution
While most switches are effortless, some medications have a "narrow therapeutic index" (NTI). This is a fancy way of saying that a tiny change in the amount of drug in your blood can be the difference between the medicine working and it being toxic-or not working at all.
If you are taking any of the following, talk to your doctor before switching manufacturers:
- Levothyroxine (for thyroid issues)
- Warfarin (blood thinner)
- Phenytoin or Carbamazepine (antiepileptics)
In the case of antiseizure meds, some patients report breakthrough seizures when switching between different generic brands. The issue isn't that the generic is "bad," but that the body has become calibrated to one specific manufacturer's formulation. If you find a generic that works perfectly, ask your pharmacist to stick with that specific manufacturer every time you refill.
Your Step-by-Step Switching Plan
Don't just swap your pills and hope for the best. Use a structured approach to ensure your health remains stable during the transition.
- Consult the Orange Book: This is the official FDA list of therapeutically equivalent drugs. Your pharmacist uses this, but you can ask them to confirm the generic is "AB-rated," meaning it's bioequivalent.
- Request a Counseling Session: Spend 5 to 10 minutes with your pharmacist. Ask about the appearance of the new pill so you aren't surprised when you open the bottle.
- Review the PPI: Read the patient package insert specifically for the inactive ingredients to rule out potential allergens.
- Keep a 14-Day Diary: For two weeks, track your symptoms. If you're on a diabetes med, monitor if your fasting glucose jumps by more than 20 mg/dL. If you're on an anticoagulant, watch for INR fluctuations over 0.5 units.
- Report Issues: If you feel the drug isn't working, don't just switch back immediately. Contact your doctor and, if necessary, use the FDA's Medication Errors Reporting Program.
Common Misconceptions About Generics
There is a lingering belief that generics take longer to work because they are "cheaper." This is a myth. Because they must be bioequivalent, the speed of absorption (the Cmax and AUC) is measured to ensure the drug hits your system at the same time the brand does. If a generic took an hour longer to work, it wouldn't be approved by the FDA.
Another common fear is that generics are made in "lower quality" facilities. In reality, generic manufacturers must follow the same Current Good Manufacturing Practice (CGMP) regulations as brand-name companies. A pill made by a generic giant like Teva or Sandoz is held to the same rigorous stability and purity standards as the most expensive brand on the market.
Will my doctor be upset if I switch to a generic?
Most doctors actually prefer generics because they know patients are more likely to stick to their medication when it's affordable. However, for NTI drugs (like certain seizure or thyroid meds), your doctor may specifically write "Dispense as Written" if they want you to stay on a brand for safety reasons.
Why does my generic pill look different every time I refill it?
Your pharmacy may switch generic suppliers based on availability or price. While the active ingredient remains the same, different manufacturers use different dyes and fillers. If this confuses you or makes you anxious, ask your pharmacist if they can source the same manufacturer consistently.
Are there any drugs that simply don't have generics?
Yes. Some very complex drugs, especially in oncology (cancer treatment) and rare diseases, still have 90%+ brand-name market share. Additionally, "biologics" (complex proteins) don't have generics; they have "biosimilars," which have a different approval process and slightly different equivalence standards.
Can a generic cause new side effects?
While rare, it is possible. This usually isn't caused by the active medicine, but by the inactive fillers or dyes. If you develop a rash or stomach upset after switching, check the ingredient list for common allergens and notify your provider.
How can I tell if a generic is truly equivalent?
Check the FDA's Orange Book. If a generic is listed as "therapeutically equivalent," it means the FDA has verified that the drug's strength, quality, and purity match the brand-name version.
Next Steps for Your Transition
If you're ready to save money, start by asking your pharmacist for a cost comparison between your current brand and the available generics. If you're switching a medication for a chronic condition like hypertension or high cholesterol, the transition is usually a non-event. However, if you're managing a complex neurological condition, schedule a brief check-in with your specialist two weeks after the switch to ensure your dosage is still hitting the mark.







Carol Yang April 27, 2026
switching to generics saved me a ton of money last year, definitely worth a try if you're on the fence!
Brittney Prince April 28, 2026
Sure, the active ingredient is the same, but who knows what's actually in those fillers?
Big Pharma probably loves it when we switch to cheap versions just so they can test different chemicals on us without us even knowing. I'll stick to the brand and keep my sanity.
Ben Jima April 29, 2026
It's great to see the focus on bioequivalence here. For anyone feeling nervous, just remember that the FDA standards are incredibly tight. If you're worried, just be transparent with your pharmacist about your concerns and keep that symptom log! You've got this!
Sharyl Foster April 30, 2026
Actually, the 4.1% variation is just an average. For some people, that 'small' gap is huge, especially with things like thyroid meds. It's kind of naive to say it's a 'seamless transition' for everyone when some of us actually feel the difference in our veins.
Jaclyn Vo May 2, 2026
OMG I literally had a panic attack the first time my pill changed from blue to white!! 😱 I thought the pharmacy messed up my whole life! It's so stressful when they just swap it without telling you first 🙄
Vijay AGarwal May 3, 2026
This is a monumental guide! In my experience, the cost difference is not just 'significant'-it is a life-saver for millions of people who would otherwise be forced to choose between food and medicine! It is absolutely heartbreaking that brand names are priced so high initially!
Andre Ojakäär May 3, 2026
everyone knows the orange book is the gold standard lol. just check the AB rating and move on. no need to overthink it. its literally just chemistry
Eric Mwiti May 5, 2026
Oh sure, because a 'confidence interval' is exactly what I need to feel totally safe while taking a pill that looks like a piece of chalk.
James Harrison May 5, 2026
It's interesting how we associate the 'brand' with quality, almost as a psychological placebo. We've been conditioned to believe the more expensive version is safer, but the reality is often just a matter of marketing and patent laws.
William Zhigaylo May 7, 2026
The lack of rigor in some of these claims is appalling. It is an absolute travesty that patients are encouraged to 'track symptoms' for two weeks rather than demanding the highest quality pharmaceutical grade products regardless of the cost. This is an insult to those of us who demand perfection in our medical care.
Anand Mehra May 8, 2026
purely a cost game. pharma companies play the market. generic is just a tool for the masses to stay barely functional
Hayley Redemption May 8, 2026
I find it quaint that people are still debating this. If you actually understand the pharmacokinetic parameters, the debate ends. The brand name is simply a luxury tax for those who enjoy the aesthetic of a specific pill color.
Michael Chukwuma May 8, 2026
I totally get why people feel anxious about it. I felt the same way when I switched my meds, so just take your time and do what feels right for you.
Beena Garud May 10, 2026
One must contemplate the ethical implications of the patent system. While the pursuit of innovation requires financial incentive, the subsequent barriers to affordable healthcare create a profound societal imbalance that only generics can partially rectify.
Karyn Tindall May 11, 2026
The absolute horror of realizing your medication has changed without warning is just too much to bear! My heart literally stopped when I saw the new shape! It is an outrage that pharmacies do this to us!